Architectural Technical Code for Hospital Clean Operating Department
Backgrond
This code is formulated to standardize the construction of hospital clean operating department, ensure the quality of medical treatment, prevent infection and guarantee medical safety.
This code is applicable to the construction and acceptance of newly built, expanded and reconstructed clean operating departments in hospitals.
The construction of hospital clean operating department must carry out the relevant national policies and technical and economic policies, achieve advanced technology, safety and applicability, economic rationality, and ensure quality.
In addition to complying with this code, the construction of hospital clean operating department should also comply with the relevant mandatory standards of the state.
Contents
- Foreword
- 1 General provisions
- 2 Terms
- 3 Classification and technical index
- 3.1 Classification
- 3.2 Technical index
- 4 Architecture
- 4.1 Layout
- 4.2 Area
- 4.3 Height
- 4.4 Doors and windows
- 4.5 Walls, ground and top
- 5 Heating, ventilation and air-conditioning
- 5.1 General provisions
- 5.2 Air purification system
- 5.3 Air-conditioning system
- 5.4 Ventilation system
- 5.5 Control system
- 6 Water supply and drainage
- 6.1 General provisions
- 6.2 Water supply
- 6.3 Drainage
- 6.4 Sewage treatment
- 7 Electrical
- 7.1 General provisions
- 7.2 Power supply
- 7.3 Lighting
- 7.4 Communication and signal
- 7.5 Automatic control
- 7.6 Grounding
- 7.7 Lightning protection
- 8 Construction and acceptance
- 8.1 General provisions
- 8.2 Construction
- 8.3 Acceptance
- Appendix A Calculation of self-purification time in clean operating room
- Appendix B Calculation of fresh air volume in clean operating department
- Appendix C Calculation of noise in clean operating room
- Appendix D Calculation of illumination in clean operating room
- Explanation of wording in this code
- List of quoted standards
1 General Provisions
1.0.1 This code is formulated to standardize the design, construction and acceptance of hospital clean operating department, improve the control ability of medical environment in hospital clean operating department.
1.0.2 This code is applicable to the design, construction and acceptance of newly built, expanded and reconstructed clean operating departments in hospitals.
1.0.3 The design, construction and acceptance of hospital clean operating department should not only comply with this code, but also comply with the relevant mandatory standards of the state.
2 Terms
2.0.1 Clean room A room where the concentration of airborne particles is controlled, and which is constructed and used in a manner to minimize the introduction, generation and retention of particles inside the room, and in which other relevant parameters, e.g. temperature, humidity and pressure, are controlled as necessary.
— ISO 14644 “Cleanrooms and associated controlled environments”
2.0.2 Clean operating room An operating room that uses air cleaning technology to reduce the airborne microbial particles and total particles in the operating environment to an acceptable level.
— GB50333-2013 “Architectural technical code for hospital clean operating department”
2.0.3 Purification air conditioning system An air conditioning system that mainly uses filtration to control the concentration of suspended dust and microorganisms in the controlled area to the required level.
— GB50333-2013 “Architectural technical code for hospital clean operating department”
2.0.4 Unidirectional flow The air flow with uniform velocity in parallel streamlines.
2.0.5 Non-unidirectional flow The air flow that is not unidirectional flow.
2.0.6 Turbulent flow The air flow that is not unidirectional flow.
Note: Turbulent flow is the term used in the earlier version of this code, which is equivalent to non-unidirectional flow in this version.
2.0.7 Local unidirectional flow The unidirectional flow in a local area.
2.0.8 Clean air ceiling The air supply device with high efficiency filter installed at the top of the operating area in the clean operating room.
2.0.9 Self-purification time The time required for the clean room to recover from a certain pollution state to the required cleanliness level after the air purification system starts to operate.
3 Classification and Technical Index
3.1 Classification
3.1.1 The clean operating rooms should be divided into four classes according to the air cleanliness level: class I, class II, class III and class IV.
3.1.2 The clean auxiliary rooms should be divided into three classes according to the air cleanliness level: class II, class III and class IV.
3.1.3 The cleanliness class of clean operating rooms and clean auxiliary rooms should comply with the provisions in Table 3.1.3.
| Class | Clean operating room | Clean auxiliary room |
|---|---|---|
| I | Special clean operating room | – |
| II | Standard clean operating room | Clean corridor, clean preparation room, clean storage room |
| III | General clean operating room | Brush hand room, anesthesia preparation room, precision instrument room, disposable item room |
| IV | Quasi clean operating room | Changing room, rest room, nurse station, preoperative and postoperative room, family waiting room |
Note: 1 The cleanliness class of clean corridor can be one level lower than the clean operating room it serves.
2 The cleanliness class of changing room can be class IV or quasi clean.
3 The cleanliness class of family waiting room can be quasi clean.
3.2 Technical Index
3.2.1 The technical index of clean operating rooms should comply with the provisions in Table 3.2.1.
| Class | Air cleanliness class | Colony count (CFU/m³) | Surface microbial count (CFU/cm²) | Sediment bacteria count (CFU/ (30min·90mm dish)) | Air change (times/h) | Section wind speed (m/s) | Temperature (°C) | Relative humidity (%) | Noise [dB(A)] | Static pressure difference (Pa) | Minimum illuminance (lx) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| I | 5 | ≤5 | ≤0.2 | ≤0.1 | – | 0.25~0.30 | 21~25 | 40~60 | ≤52 | ≥10 | 300 |
| II | 6 | ≤200 | ≤3 | ≤2 | ≥40 | – | 22~25 | 40~60 | ≤52 | ≥8 | 300 |
| III | 7 | ≤500 | ≤5 | ≤4 | ≥30 | – | 22~25 | 40~60 | ≤52 | ≥6 | 300 |
| IV | 8 | ≤1000 | ≤10 | ≤6 | ≥20 | – | 22~25 | 40~60 | ≤55 | ≥5 | 300 |
Note: 1 For class I clean operating room, the air cleanliness class at 0.5m above the operating table should be class 5.
2 The colony count is the maximum allowable average concentration.
3 The noise is the maximum allowable value.
3.2.2 The technical index of clean auxiliary rooms should comply with the provisions in Table 3.2.2.
| Class | Air cleanliness class | Colony count (CFU/m³) | Surface microbial count (CFU/cm²) | Sediment bacteria count (CFU/ (30min·90mm dish)) | Air change (times/h) | Temperature (°C) | Relative humidity (%) | Noise [dB(A)] | Static pressure difference (Pa) | Minimum illuminance (lx) |
|---|---|---|---|---|---|---|---|---|---|---|
| II | 6 | ≤200 | ≤3 | ≤2 | ≥30 | 22~26 | 40~60 | ≤52 | ≥8 | 200 |
| III | 7 | ≤500 | ≤5 | ≤4 | ≥20 | 22~26 | 40~60 | ≤52 | ≥6 | 200 |
| IV | 8 | ≤1000 | ≤10 | ≤6 | ≥15 | 22~26 | 40~60 | ≤55 | ≥5 | 200 |
Note: 1 The colony count is the maximum allowable average concentration.
2 The noise is the maximum allowable value.
3 For class IV clean auxiliary rooms, the air change can be 10~15 times/h if mechanical ventilation is adopted.
4 Architecture
4.1 Layout
4.1.1 The clean operating department should be self-contained and divided into clean area and non-clean area. The clean area should include clean operating rooms, preparation room, isolation operating room, brush hand room, storage room for disposable items, anesthesia preparation room, resuscitation room, nurse station, etc. The non-clean area should include duty room, office, restroom, changing room, preoperative and postoperative room, family waiting room, etc.
4.1.2 The layout of clean operating department should comply with the following provisions:
1 The clean area and non-clean area should be clearly divided, with separate entrances and exits, and should not be crossed.
2 The layout should meet the requirements of functional process and clean and dirty partition, and achieve reasonable flow of people, logistics, clean and dirty.
3 The evacuation exit should be set according to the requirements of fire protection design.
4.1.3 The clean corridor should be set between clean operating rooms, and the clean corridor should not be crossed by non-clean flow.
4.1.4 The isolation operating room should be set separately, with separate entrance and exit, and should not share the clean corridor with other operating rooms.
4.1.5 The changing room should be divided into two areas: clean and non-clean, with a buffer zone in between.
4.1.6 The preoperative and postoperative room and family waiting room should be set in the non-clean area.
4.1.7 The storage room for disposable items should be set in the clean area, and the items should be transported by a dedicated elevator or lift.
4.2 Area
4.2.1 The area of clean operating room should comply with the provisions in Table 4.2.1.
| Class | Minimum area (m²) |
|---|---|
| I | 40 |
| II | 35 |
| III | 30 |
| IV | 25 |
Note: For class I and class II clean operating rooms, the area can be appropriately increased when necessary.
4.2.2 The area of clean auxiliary rooms should comply with the provisions in Table 4.2.2.
| Room | Minimum area per operating room (m²) |
|---|---|
| Brush hand room | 6 |
| Anesthesia preparation room | 10 |
| Precision instrument room | 10 |
| Disposable item room | 10 |
| Clean corridor | Calculated according to the number of operating rooms and layout |
Note: The area of clean corridor should be determined according to the functional requirements.
4.3 Height
4.3.1 The clear height of clean operating room should be 2.8~3.0m.
4.3.2 The clear height of clean auxiliary rooms should be 2.6~2.8m.
4.4 Doors and Windows
4.4.1 The doors of clean operating room should be automatic airtight doors, and the width of the door opening should not be less than 1.4m.
4.4.2 The doors of clean auxiliary rooms should be airtight doors.
4.4.3 The windows of clean operating room and clean auxiliary rooms should be double-layer airtight windows, and the window sill height should not be less than 1.0m.
4.4.4 The clean operating department should not set openable windows.
4.5 Walls, Ground and Top
4.5.1 The walls, ground and top of clean operating room and clean auxiliary rooms should be smooth, seamless, wear-resistant, non-glare, easy to clean and disinfect.
4.5.2 The ground of clean operating room should be anti-static.
4.5.3 The junction of walls, ground and top should be arc-shaped.
4.5.4 The walls should be equipped with anti-collision facilities.
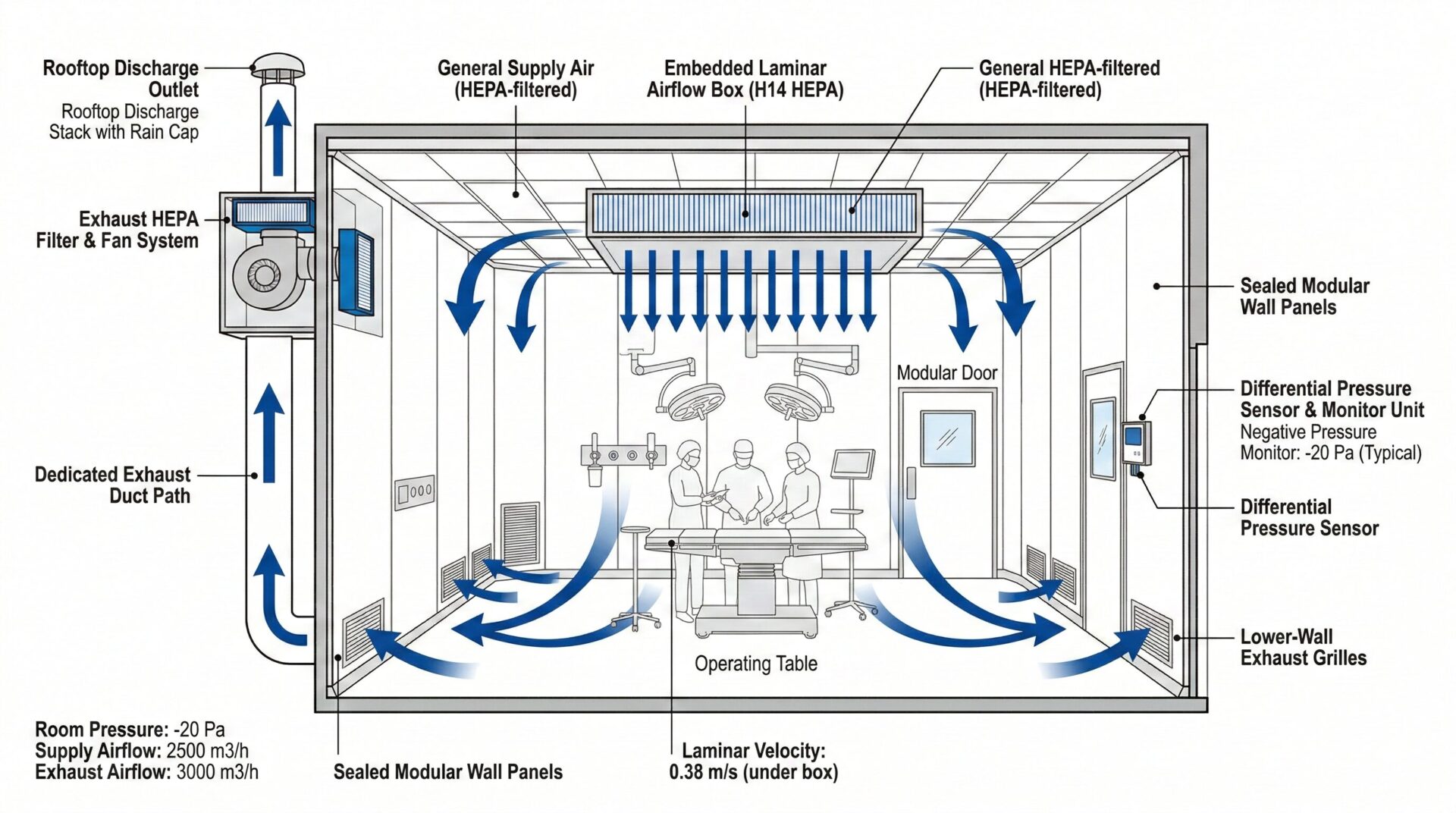
5 Heating, Ventilation and Air-Conditioning
5.1 General Provisions
5.1.1 The clean operating department should adopt purification air conditioning system.
5.1.2 The purification air conditioning system should have independent fresh air system.
5.1.3 The air purification system should be set separately for each clean operating room.
5.1.4 The air conditioning system should have automatic control and regulation functions.
5.1.5 The heating, ventilation and air-conditioning system should ensure the temperature, humidity, pressure difference, air change and cleanliness in the clean area meet the requirements.
5.1.6 The noise of the air conditioning system should meet the requirements.
5.2 Air Purification System
5.2.1 The air purification system should adopt three-level filtration: primary, medium and high efficiency filters.
5.2.2 The high efficiency filter should be set at the air supply outlet.
5.2.3 The air supply outlet of class I clean operating room should adopt clean air ceiling, and the size should not be less than 2.4m×2.6m.
5.2.4 The air supply outlet of class II and class III clean operating room should adopt high efficiency filter air supply outlet, evenly arranged above the operating area.
5.2.5 The air supply outlet of class IV clean operating room can adopt medium efficiency filter air supply outlet.
5.2.6 The return air inlet should be set at the lower part of the wall, not less than 0.1m from the ground.
5.2.7 The exhaust air outlet should be set at the top or upper part of the wall.
5.2.8 The fresh air inlet should be set outdoors, not less than 3m from the ground, and away from pollution sources.
5.2.9 The air duct should be made of galvanized steel plate or stainless steel plate, and the inner surface should be smooth.
5.2.10 The air duct should be airtight and easy to clean.
5.3 Air-Conditioning System
5.3.1 The air-conditioning system should adopt fan coil unit or fresh air + fan coil unit.
5.3.2 The cooling source should adopt chilled water or direct expansion.
5.3.3 The heating source should adopt hot water or electric heating.
5.3.4 The air-conditioning system should have humidification and dehumidification functions.
5.3.5 The air-conditioning system should have temperature and humidity automatic control.
5.4 Ventilation System
5.4.1 The ventilation system should ensure the pressure difference between rooms.
5.4.2 The pressure in clean operating room should be higher than adjacent rooms.
5.4.3 The pressure gradient should be from high to low: clean operating room > clean corridor > auxiliary room > non-clean area.
5.4.4 The isolation operating room should adopt negative pressure or positive pressure according to the requirements.
5.4.5 The exhaust air system should be set for rooms with harmful gases.
5.5 Control System
5.5.1 The control system should include temperature, humidity, pressure difference, air change, cleanliness monitoring and alarm.
5.5.2 The control system should have automatic and manual modes.
5.5.3 The control system should have energy-saving measures.
6 Water Supply and Drainage
6.1 General Provisions
6.1.1 The water supply and drainage system should meet the requirements of medical hygiene and safety.
6.1.2 The water quality should meet the standard of medical water.
6.1.3 The drainage should be smooth and prevent backflow.
6.2 Water Supply
6.2.1 The clean operating room should be equipped with medical purified water.
6.2.2 The brush hand sink should be non-touch type.
6.2.3 The water supply pipe should be made of stainless steel or copper.
6.2.4 The water supply system should have disinfection facilities.
6.3 Drainage
6.3.1 The drainage system should have anti-odor and anti-backflow measures.
6.3.2 The floor drain should be set in the clean operating room, with water seal.
6.3.3 The sewage should be treated before discharge.
6.4 Sewage Treatment
6.4.1 The sewage from clean operating department should be treated to meet the discharge standard.
6.4.2 The sewage treatment facilities should be set according to the scale.
7 Electrical
7.1 General Provisions
7.1.1 The electrical system should ensure safety, reliability and meet medical requirements.
7.1.2 The power supply should be dual circuit or equipped with emergency power.
7.1.3 The lighting should meet the requirements of illuminance and no shadow.
7.1.4 The communication and signal system should be complete.
7.1.5 The automatic control system should be reliable.
7.1.6 The grounding and lightning protection should be reliable.
7.2 Power Supply
7.2.1 The power supply load should be class I or class II.
7.2.2 The operating room should have isolated power supply system.
7.2.3 The emergency power should start within 15s.
7.3 Lighting
7.3.1 The illuminance of operating table should be 40000~160000lx.
7.3.2 The lighting should be shadowless.
7.3.3 The auxiliary lighting should be set.
7.3.4 The lighting fixtures should be easy to clean and disinfect.
7.4 Communication and Signal
7.4.1 The operating room should have intercommunication system.
7.4.2 The call system should be set.
7.4.3 The monitoring system should be set.
7.5 Automatic Control
7.5.1 The automatic control system should include air conditioning control, lighting control, etc.
7.5.2 The system should have monitoring and alarm functions.
7.6 Grounding
7.6.1 The grounding system should be TN-S system.
7.6.2 The operating room should have equipotential grounding.
7.6.3 The grounding resistance should meet the requirements.
7.7 Lightning Protection
7.7.1 The building should have lightning protection measures.
7.7.2 The electrical equipment should have surge protection.
8 Construction and Acceptance
8.1 General Provisions
8.1.1 The construction should comply with the design documents and this code.
8.1.2 The materials and equipment should meet the requirements.
8.1.3 The construction should be carried out by qualified units.
8.2 Construction
8.2.1 The construction sequence should be reasonable.
8.2.2 The airtightness should be ensured.
8.2.3 The anti-static ground should be constructed according to the requirements.
8.2.4 The pipeline installation should be concealed.
8.3 Acceptance
8.3.1 The acceptance should include single item acceptance and comprehensive acceptance.
8.3.2 The cleanliness, temperature, humidity, pressure difference, noise, illuminance should be tested.
8.3.3 The acceptance should be carried out by qualified units.
Appendix A Calculation of Self-Purification Time in Clean Operating Room
The formula for self-purification time t is:
t = (1/n) ln (C0 / C)
where n is air change rate (times/h), C0 is initial concentration, C is final concentration.
Appendix B Calculation of Fresh Air Volume in Clean Operating Department
The fresh air volume Q is:
Q = max(Q1, Q2, Q3)
where Q1 is for personnel, Q2 for heat, Q3 for humidity.
Appendix C Calculation of Noise in Clean Operating Room
Noise level L = 10 log (P / P0)
where P is sound power, P0 is reference.
Appendix D Calculation of Illumination in Clean Operating Room
Illuminance E = Φ / A
where Φ is luminous flux (lm), A is area (m²).
Explanation of Wording in This Code
Mandatory provisions use “must”, recommended use “should”, permissive use “may”.
List of Quoted Standards
- GB 50016 Code for fire protection design of buildings
- GB 50045 Code for fire protection design of tall buildings
- GB 50116 Code for design of automatic fire alarm system
- GBJ 84 Specification for design of sprinkler systems
- GB 50084 Code for design of sprinkler systems
- GB 50174 Code for design of electronic computer room
- GB/T 16292 Test method for airborne particles in clean room (zone) of the pharmaceutical industry
- GB/T 16293 Test method for airborne microbes in clean room (zone) of the pharmaceutical industry
- GB/T 16294 Test method for settling microbes in clean room (zone) of the pharmaceutical industry
- JGJ 49 Technical code for construction of hospital buildings
- JGJ 91 Code for design of scientific experiment buildings
- YY/T 0186 General technical requirements for medical clean benches
- YY 0033 Good manufacture practice for sterile medical devices